Mounjaro vs Wegovy UK: A Clinical Comparison

In the field of medical weight management in the UK, two treatments are frequently discussed: Mounjaro and Wegovy. Both are once-weekly injectable medications, but a key distinction lies in their clinical trial results. Evidence from head-to-head studies indicates that Mounjaro (tirzepatide) has been associated with greater average weight loss compared to Wegovy (semaglutide).
This difference is attributed to Mounjaro's dual-action mechanism, which targets two distinct gut hormones involved in appetite and metabolic regulation.
An Evidence-Based UK Comparison
For individuals exploring weight management options, understanding the clinical differences between Mounjaro and Wegovy is crucial. While both are indicated for adults with obesity or those who are overweight with related health conditions, their pharmacological actions and clinical outcomes differ.
This guide provides an evidence-based overview of these two treatments within the UK healthcare context. We will examine their mechanisms of action, compare results from major clinical trials, and outline the pathways for access, whether through the NHS or private clinics. The objective is to provide clear, factual information to facilitate informed discussions with a healthcare provider.
Key Differences at a Glance
The primary distinction lies in their active ingredients and mechanisms of action. Mounjaro's ability to engage two hormonal pathways is the main reason it has demonstrated higher average weight loss in clinical studies. Wegovy, which targets a single hormone, remains a highly effective and established option for weight management.
The image below illustrates this fundamental difference.
As shown, Mounjaro interacts with both GIP and GLP-1 receptors, whereas Wegovy focuses solely on the GLP-1 receptor.
Mounjaro's dual agonism of both GLP-1 and GIP receptors is thought to be responsible for its enhanced effects on appetite suppression and metabolic function when compared to single-agonist medications like Wegovy.
This pharmacological difference translates into clinical outcomes. Head-to-head clinical trials consistently report a superior weight loss outcome for Mounjaro. One major study found that participants on the highest dose of tirzepatide lost an average of 20.9% of their body weight over 72 weeks, compared to 14.9% for those on semaglutide.
To see a broader view, you can review the detailed statistics on weight loss injections and understand the data behind these figures.
For a quick summary, the table below breaks down the essential features of each medication.
Mounjaro vs Wegovy At a Glance
This table provides a simple, side-by-side view of the core differences between Mounjaro and Wegovy, based on available clinical information.
| Feature | Mounjaro (Tirzepatide) | Wegovy (Semaglutide) |
|---|---|---|
| Active Ingredient | Tirzepatide | Semaglutide |
| Mechanism of Action | Dual GIP/GLP-1 Receptor Agonist | GLP-1 Receptor Agonist |
| UK Indication | Weight Management & Type 2 Diabetes | Weight Management |
| Administration | Once-weekly subcutaneous injection | Once-weekly subcutaneous injection |
| Avg. Weight Loss | Up to 22.5% of body weight in clinical trials | Around 15% of body weight in clinical trials |
Ultimately, while both medications are administered in the same manner and serve a similar purpose, their underlying pharmacology and the results observed in clinical trials are notably different.
Understanding the Science Behind Each Medication
To fully compare Mounjaro and Wegovy, it is necessary to examine their underlying mechanisms of action. Both medications interact with the body’s incretin system, which naturally regulates appetite and metabolism. This interaction is central to their efficacy in weight management.
The foundation for both treatments is a gut hormone called glucagon-like peptide-1 (GLP-1). The body releases GLP-1 following a meal, which signals satiety to the brain, slows gastric emptying, and supports blood sugar regulation. The cumulative effect is reduced hunger and lower caloric intake.

Wegovy: A Potent GLP-1 Agonist
Wegovy’s active ingredient, semaglutide, is classified as a GLP-1 receptor agonist. This means it is engineered to mimic the effects of the natural GLP-1 hormone but with a significantly longer duration of action in the body.
By activating GLP-1 receptors in the brain and gut, Wegovy enhances signals of satiety and fullness. This physiological response typically leads to reduced food intake and subsequent weight loss. This single-hormone approach has been a significant advancement in medical weight management, as it directly targets a primary pathway controlling hunger.
Mounjaro: The Dual-Action Difference
Mounjaro, with its active ingredient tirzepatide, represents a different pharmacological approach. It is the first in a class of medications known as dual GIP and GLP-1 receptor agonists, which is the fundamental difference between it and Wegovy.
Mounjaro not only mimics GLP-1 but also imitates a second incretin hormone called glucose-dependent insulinotropic polypeptide (GIP). Like GLP-1, GIP is released after eating and plays a role in energy balance and fat metabolism.
This dual-agonist function is what distinguishes Mounjaro. By targeting two separate hormone pathways simultaneously, it is believed to create a more potent, synergistic effect on appetite control and metabolic health. Evidence suggests this combination may lead to greater appetite suppression and improved fat metabolism compared to targeting GLP-1 alone. You can find a more detailed explanation in our guide on how Mounjaro works.
The key takeaway is that while Wegovy effectively mimics one powerful appetite-regulating hormone, Mounjaro targets two. This dual action is the scientific basis for the greater average weight loss observed in head-to-head clinical trials.
Activating both GIP and GLP-1 receptors appears to have a more profound impact on the body’s metabolic system, influencing glucose and lipid metabolism. This scientific foundation explains why clinical trial data, which we will explore next, often show a difference in outcomes between the two treatments.
Comparing Efficacy With Head-to-Head Trial Data
A primary consideration for any weight management treatment is its efficacy. For Mounjaro and Wegovy, extensive clinical trial data is available. While both have demonstrated significant effectiveness, head-to-head studies consistently show that Mounjaro's dual-action mechanism is associated with greater average weight loss.
This section examines the evidence from the landmark clinical trial programmes: the SURMOUNT trials for tirzepatide (Mounjaro) and the STEP programme for semaglutide (Wegovy). Comparing their results provides a clear, factual picture of their relative efficacy.
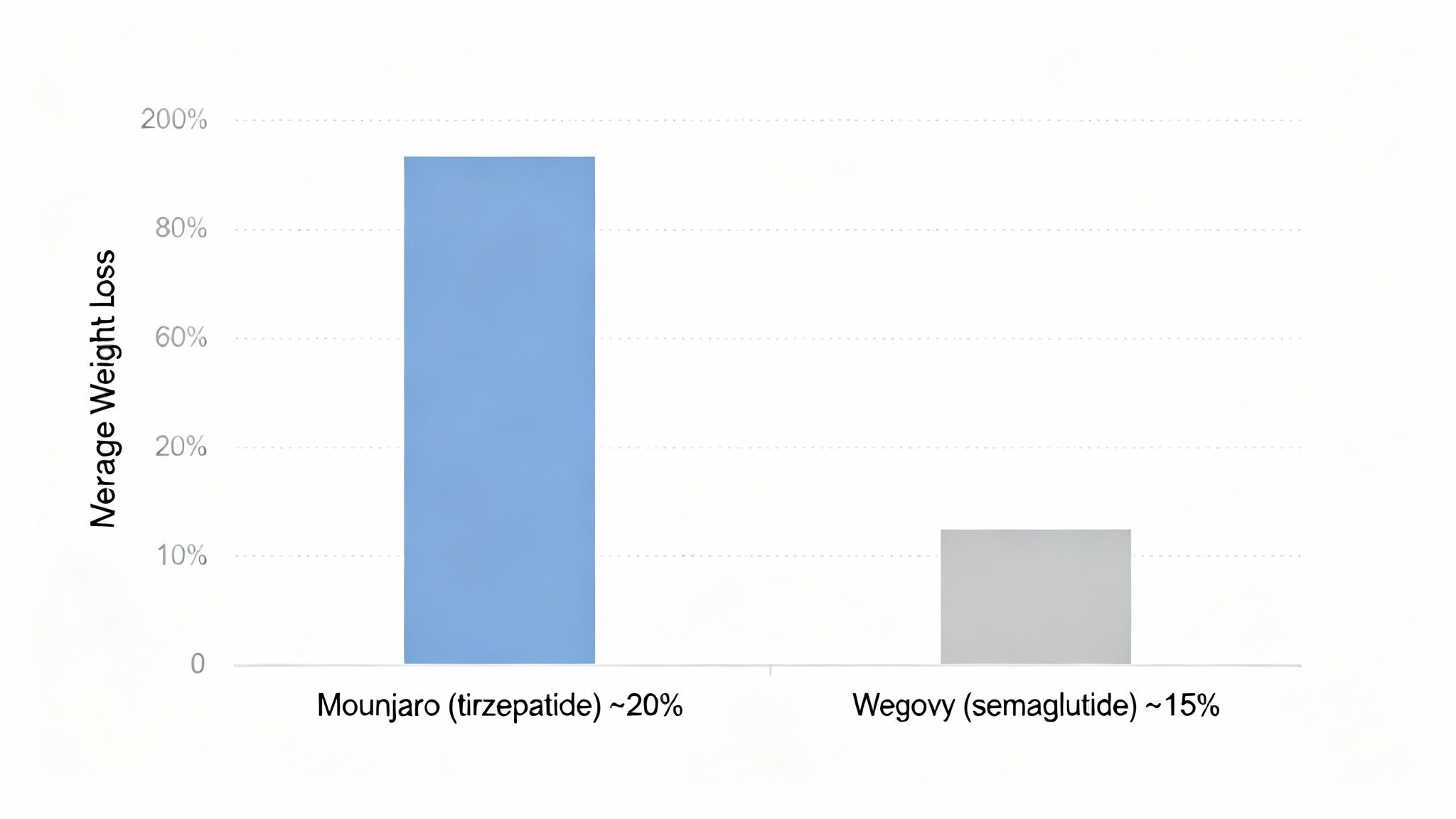
The Primary Endpoint: Average Weight Loss
The primary objective of these clinical trials was to measure the mean percentage of body weight lost by participants over a defined period, typically 68 to 72 weeks. This "primary endpoint" is the clearest indicator of a medication's efficacy.
Direct comparison trials provide the most definitive data. In a key head-to-head study, participants receiving the highest dose of Mounjaro achieved significantly greater weight loss than those on the highest dose of Wegovy.
Participants on Mounjaro (tirzepatide) lost an average of 20.9% of their baseline body weight over 72 weeks. This compared to an average loss of 14.9% for those taking Wegovy (semaglutide) in the same trial.
This is a clinically meaningful difference. In practical terms, for an individual weighing 100kg (approx. 15 stone 10 lbs), this could equate to an additional 6kg (almost one stone) of weight loss with Mounjaro compared to Wegovy over the study duration.
Proportions of Significant Weight Loss
Beyond averages, it is informative to examine the proportion of participants who achieved major weight loss milestones. The trials tracked the percentage of participants losing at least 5%, 10%, 15%, 20%, and 25% of their body weight.
Here again, the data demonstrates a greater effect for Mounjaro. A higher percentage of individuals taking tirzepatide reached these significant weight loss targets.
- Losing over 20% of Body Weight: Approximately 57% of participants on the highest Mounjaro dose achieved this, compared to about 36% of those on Wegovy.
- Losing over 25% of Body Weight: The difference becomes more pronounced at this threshold, with 36% of Mounjaro users reaching this milestone, versus 19% of Wegovy users.
These figures indicate that while both are effective treatments, Mounjaro appears to offer a higher probability of achieving a more substantial degree of weight reduction for a larger proportion of users. If you are interested in the broader context of these treatments, you can read also our guide to weight loss injections in the UK.
Impact on Cardiometabolic Health Markers
Weight management is intrinsically linked to improving overall health. Clinical trials for both Mounjaro and Wegovy measured key secondary outcomes related to cardiometabolic health, with positive results observed for both.
However, consistent with its greater effect on weight, Mounjaro also demonstrated superior improvements across several of these health markers in head-to-head comparisons.
Key Cardiometabolic Improvements:
- Waist Circumference: Mounjaro users experienced a greater average reduction in waist circumference, an important indicator of visceral fat.
- Blood Pressure: Both systolic and diastolic blood pressure readings showed more significant reductions in the Mounjaro group.
- Lipids (Cholesterol): Participants on Mounjaro showed greater improvements in lipid profiles, including lower triglycerides and LDL cholesterol.
- Blood Sugar Control: Both medications are highly effective at improving glycemic control. The mechanisms of Mounjaro and Wegovy have a profound impact on this, a key point also discussed when looking into any legitimate blood sugar product for managing levels. In trials, Mounjaro often demonstrated a slight advantage in reducing HbA1c, a measure of long-term blood sugar levels.
In summary, the clinical evidence from direct trials is consistent. While Wegovy is a highly effective treatment that produces significant weight loss, the data indicates Mounjaro is statistically superior in terms of average percentage of weight lost and the proportion of individuals achieving higher tiers of weight reduction. This advantage also extends to greater improvements in key markers of metabolic health, providing a solid evidence base for clinicians and patients in the UK to discuss treatment options.
Getting Started: Side Effects, Dosing, and How to Use the Pens
Initiating treatment with either Mounjaro or Wegovy involves understanding the practical aspects of the medication: potential side effects, the dosing schedule used to manage them, and the correct use of the injection pen.
As both treatments work by influencing the digestive system, their side effect profiles are very similar. The most commonly reported adverse events are gastrointestinal in nature, such as nausea, diarrhoea, constipation, and abdominal discomfort, particularly during the initial phase of treatment. This is an expected consequence of the medication slowing gastric emptying, a key part of its mechanism of action.
For most individuals, these side effects are transient. They are typically most pronounced when starting the treatment or after a dose escalation and tend to subside as the body acclimatises. Lifestyle adjustments, such as eating smaller, more frequent meals and avoiding high-fat foods, can help mitigate these effects.
Why “Start Low, Go Slow” is Key
To facilitate adaptation, both Mounjaro and Wegovy employ a strict dose titration schedule. This means treatment begins at a low starting dose and is gradually increased over several months under clinical supervision.
This gradual approach is crucial for both safety and tolerability. It allows the body time to adjust, which significantly reduces the intensity of potential side effects. Adherence to the prescribed titration plan is essential for a positive treatment experience.
Each medication has a specific schedule designed to balance efficacy with tolerability.
Wegovy Dosing Schedule (UK):
- Month 1: 0.25 mg once a week
- Month 2: 0.5 mg once a week
- Month 3: 1.0 mg once a week
- Month 4: 1.7 mg once a week
- Month 5 onwards: 2.4 mg (maintenance dose) once a week
Mounjaro Dosing Schedule (UK):
- Weeks 1-4: 2.5 mg once a week
- Weeks 5-8: 5.0 mg once a week
- Subsequently, the dose may be increased in 2.5 mg increments every four weeks, based on clinical response and tolerability. The maximum maintenance dose is 15 mg.
The principle for both is identical: "start low, go slow." This gradual dose escalation is the most important strategy for managing side effects and ensuring the body adapts smoothly to the treatment.
Using the Pen Devices
Mounjaro and Wegovy are administered via a once-weekly subcutaneous injection using a pre-filled pen. A subcutaneous injection delivers the medicine into the fatty tissue just beneath the skin, typically in the abdomen, thigh, or upper arm.
The pens are designed for ease of use at home. They feature a fine, hidden needle to make the process quick and minimally uncomfortable for most users. The injection is typically administered with a simple push-button mechanism.
While the basic process is similar, there are minor differences between the pen designs. The Mounjaro pen is a single-use device with an integrated, self-locking needle. The Wegovy FlexTouch pen is also a straightforward device with a built-in needle. The prescribing healthcare provider will provide full training on the correct and safe use of the specific pen.
How to Access Mounjaro and Wegovy in the UK
In the UK, Mounjaro and Wegovy are prescription-only medications available via two distinct pathways: the National Health Service (NHS) or a private healthcare provider.
Understanding these routes is essential for anyone considering these treatments. The choice often depends on eligibility criteria, urgency, and personal circumstances. Both pathways are regulated to prioritise patient safety but operate with different access criteria and timelines.

The NHS Pathway
Accessing Mounjaro or Wegovy on the NHS requires meeting strict guidelines set by the National Institute for Health and Care Excellence (NICE). These criteria ensure the medications are provided to patients with the greatest clinical need within the NHS framework.
Eligibility involves a multi-step assessment based on a complete clinical profile.
Typical NHS Eligibility Criteria:
- A high BMI: Usually a BMI of 30 kg/m² or more. For individuals from certain ethnic backgrounds with a higher risk of comorbidities, this threshold may be lowered to 27 kg/m².
- Weight-related health conditions: At least one comorbidity, such as type 2 diabetes, prediabetes, hypertension, or sleep apnoea, must be present.
- Specialist Service Enrolment: The prescription must be initiated by a specialist, tier 3 weight management service. This requires a GP referral and often involves a significant waiting period for an initial consultation.
This structured approach ensures the medication is part of a comprehensive, medically supervised programme. However, high demand and limited capacity in these specialist services can result in lengthy delays.
The Private Healthcare Pathway
The private sector offers a more direct route for many individuals. Regulated digital health clinics provide an alternative for those who meet the prescribing criteria but may not qualify under the strict NHS guidelines or wish to avoid long waiting times.
The process with a private clinic is designed to be thorough yet efficient, with patient safety as the primary concern. It typically begins with a detailed online consultation, which is reviewed by a UK-based clinician to determine suitability for treatment.
This pathway addresses the gap between public demand and the limited capacity of the NHS. A recent UCL study estimated that 1.6 million adults in England, Wales, and Scotland used weight loss drugs like Mounjaro and Wegovy between early 2023 and early 2024. This figure significantly exceeds the NHS England's target of treating 220,000 people over three years, highlighting the substantial role of private provision. You can read the full research on this surge in demand for more details.
The private route is not a shortcut around safety; it is a regulated alternative for eligible individuals, offering faster access to a clinician's assessment, prescription, and the ongoing support required for a weight management programme.
Following clinician approval, the medication is dispatched from a registered pharmacy directly to the patient's home. This model combines medical rigour with convenience. For the latest on available doses, you can learn about the arrival of higher-strength Wegovy in the UK.
Regardless of the pathway, the decision to prescribe Mounjaro or Wegovy rests with a qualified clinician who will assess an individual's health profile to determine the most appropriate and safe course of action.
So, Which Treatment is Right for You?
The choice between Mounjaro and Wegovy is a clinical decision, not a matter of personal preference. It should be made in consultation with a qualified clinician who can assess your individual health profile, medical history, and treatment goals.
The decision is based on nuanced clinical factors. A clinician will consider the different mechanisms of action in the context of your specific circumstances.
Your Personal Health Story Matters
For instance, if a patient has previously had a suboptimal response to a GLP-1-only medication, a clinician might consider Mounjaro. Its dual GIP/GLP-1 receptor agonism could offer a more effective therapeutic pathway.
Conversely, for a patient new to this class of medication, a clinician might discuss Wegovy, which has a longer-established evidence base specifically for weight management as a single-agonist therapy.
It is important to remember that neither Mounjaro nor Wegovy is a standalone solution. Their efficacy is maximised when used as part of a comprehensive plan that includes dietary modifications, increased physical activity, and ongoing behavioural support.
Market and Prescribing Trends
Patient and clinician preferences are shaping prescribing patterns. In the UK's private market, Mounjaro has become a widely prescribed GLP-1 medication. It currently accounts for a 79% share, with Wegovy at 20%, according to recent private clinic data. This trend likely reflects the compelling weight loss results from its clinical trials.
What's Your Next Step?
The primary goal is sustainable, long-term health improvement. While medications like Mounjaro and Wegovy are valuable tools, a holistic approach is essential. This includes incorporating regular physical activity. For those starting out, it can be useful to identify effective tools, such as the best cardio machines for weight loss.
The definitive next step is a medical consultation. A clinician is the only professional qualified to evaluate your complete health profile, discuss the potential benefits and risks of each medication, and help determine the safest and most effective plan to achieve your health goals.
Frequently Asked Questions
Here are answers to some of the most common questions about Mounjaro and Wegovy, based on current clinical understanding.
Can I Switch From Wegovy to Mounjaro?
Yes, switching from Wegovy to Mounjaro is possible but must be managed by a clinician. A healthcare professional is the only person who can determine if this is a safe and appropriate course of action for you.
If a switch is deemed suitable, the clinician will provide a specific "washout" period and a new titration schedule. This ensures a safe transition and helps minimise side effects. You should never attempt to switch these medications without direct medical supervision.
Are These Medications a Lifelong Commitment?
Weight management is often a long-term process. These medications are most effective during active treatment. Clinical trials indicate that upon cessation of treatment, weight regain is common unless sustainable lifestyle changes have been successfully implemented.
For this reason, these medications are often considered long-term therapies for maintaining a healthier weight. The duration of treatment is a clinical decision made in consultation between the patient and their clinician.
What Are the Key Differences in Contraindications?
The contraindications for Mounjaro and Wegovy are very similar, as they are both in the GLP-1 agonist class. Neither medication should be used by individuals with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
A thorough review of your full medical history is a critical part of the consultation. Any history of pancreatitis or severe gastrointestinal disease, for example, requires careful consideration before initiating either treatment.
If I Am Eligible for Both, How Do I Decide?
If a clinician determines that you are a suitable candidate for both Mounjaro and Wegovy, the decision often depends on your individual health goals and medical history. Given that Mounjaro's dual-action mechanism is associated with higher average weight loss in clinical trials, it might be considered if maximising weight reduction is a primary objective.
Alternatively, a clinician might discuss Wegovy if its longer track record specifically for weight management is a more relevant factor. Ultimately, the most appropriate choice is made collaboratively with your healthcare provider based on a comprehensive clinical assessment.
Your health journey is unique. At Trim, our UK-registered clinicians are here to help you understand your options and create a safe, effective, and medically supervised weight management plan. Start your free consultation today at https://gettrim.co.uk.
Accuracy & Review Notice
This article is accurate as of 09/02/2026 and reflects the clinical guidance and evidence available at the time of publication. Content has been checked and verified by Roger Compton (GPhC: 2082993). Medical guidance may change over time; readers should seek personalised advice from a qualified healthcare professional.
